Ethical notice: This article is for educational purposes only and does not replace medical advice. Treatment decisions must be individualized. If peritoneal metastases are suspected or confirmed, evaluation in an experienced Peritoneal Surface Malignancy (PSM) center is strongly recommended.
Introduction
Blue March is the global awareness month for colon cancer. Beyond prevention and early detection, there is a topic that deserves far more clarity: peritoneal metastases (often labeled “peritoneal carcinomatosis”). Peritoneal spread is not rare in colon cancer, and reported incidence varies by population and detection strategy.[1] The real-world burden is frequently underestimated because imaging has well-known limitations, particularly for small implants.[1]
This matters because once colon cancer involves the peritoneum, the strategy changes. You need better staging, better patient selection, and—most importantly—avoid improvised decisions that can increase morbidity and reduce future therapeutic options.
Recommended reading on Peritoneo.life (optional):
* Incidental peritoneal metastases in emergency surgery (a decision framework): https://peritoneo.life/incidental-peritoneal-metastases-emergency-surgery/
* Why disease volume matters (PCI explained): https://peritoneo.life/carcinomatose-peritoneal-quantidade-tratamento/
* ctDNA in colorectal cancer (ASCO GI 2025 insights): https://peritoneo.life/circulating-tumor-dna-in-colorectal-cancer-asco-gi-2025-insights/
Key takeaways
- Peritoneal involvement is associated with worse survival compared with isolated non-peritoneal metastases in robust comparative analyses under systemic therapy.[2]
- In ARCAD-based summaries, patients with isolated peritoneal metastases had median overall survival around 16–17 months on systemic therapy.[2]
- CT imaging can miss small-volume peritoneal disease; for lesions <0.5 cm, sensitivity can be extremely low in lesion-based analyses, and CT-PCI tends to underestimate surgical PCI.[3]
- The Dutch randomized trial by Verwaal (2003) was a historical turning point for cytoreductive surgery (CRS) + HIPEC compared to standard therapy of that era.[4]
- PRODIGE 7 showed no overall survival benefit from adding short-duration, high-dose oxaliplatin HIPEC to CRS, reinforcing that high-quality CRS is the cornerstone in selected patients.[5]
- International consensus (PSOGI) strongly recommends against short-duration, high-dose oxaliplatin HIPEC and favors Mitomycin C “Dutch protocol” in selected settings, with HIPEC remaining a conditional option after CRS.[6]
Colon cancer and the risk of peritoneal metastases
Peritoneal metastases occur when tumor cells implant on peritoneal surfaces and grow across the abdominal cavity. This pattern has distinct biology and clinical behavior compared with other metastatic sites.[1] In practice, “metastatic disease” is not a single category—the metastatic site changes prognosis, staging accuracy, and treatment options.
Why peritoneal metastases have worse outcomes
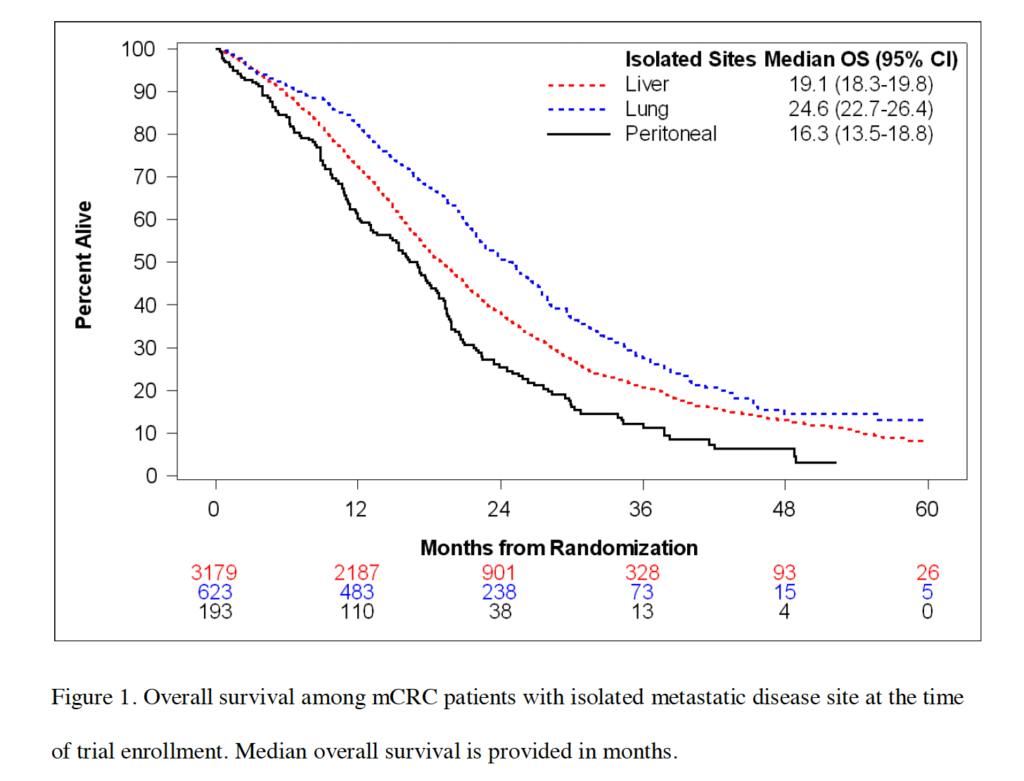
In an individual patient data analysis from ARCAD trials, outcomes for isolated non-peritoneal metastases were better than for isolated peritoneal metastases (adjusted hazard ratio favoring non-peritoneal disease).[2] Clinically, this aligns with what most multidisciplinary teams see: peritoneal involvement often behaves as a higher-risk phenotype under systemic therapy alone.
A practical explanation discussed in the literature is that peritoneal metastases may respond less effectively to systemic therapy due to locoregional disease characteristics and frequent “hidden” tumor burden underestimated by staging.[1],[3] That is why the key question is not only “which chemotherapy?” but “which full strategy?”, including structured staging and surgery when appropriate.
Imaging and staging challenges: where decisions often go wrong
Peritoneal metastases are often underdiagnosed early. CT performance depends heavily on lesion size; for implants <0.5 cm, sensitivity can be very low in lesion-based analyses, and CT-based PCI tends to underestimate the true surgical PCI.[3] Clinically, this explains a common and dangerous scenario: imaging appears “limited,” yet operative staging reveals substantially higher disease burden.
Practical implication: when peritoneal disease is suspected, staging should be high-rigor and structured, ideally with a team experienced in PSM, because understaging changes eligibility, risk, and the overall treatment plan.[1],[3]
If you want a simple explanation of why “volume matters,” read the PCI primer on Peritoneo.life:
https://peritoneo.life/carcinomatose-peritoneal-quantidade-tratamento/
The historical timeline: from palliation to CRS + HIPEC
For many years, peritoneal metastases from colon cancer were managed predominantly with palliative intent. The randomized trial by Verwaal (2003) changed the field by comparing standard therapy of the time against CRS + HIPEC and demonstrating improved survival in the surgical arm.[4] This helped solidify a principle that remains true today: outcomes depend on patient selection and the ability to achieve complete macroscopic cytoreduction.
What the PRODIGE 7 era really taught us
PRODIGE 7 tested whether adding short-duration, high-dose oxaliplatin HIPEC to CRS improved outcomes. The conclusion was clear: no overall survival benefit for that HIPEC strategy.[5]
The modern interpretation is not “HIPEC never.” It is: CRS quality is non-negotiable; HIPEC is not a single entity; and regimen details matter. In real life, this shifts the focus toward center experience, selection, and a thoughtful approach to technique and perioperative pathways.
HIPEC today: what international consensus is actually saying
PSOGI consensus guidance for colorectal peritoneal metastases is pragmatic. It strongly recommends against short-duration, high-dose oxaliplatin HIPEC and favors Mitomycin C using the Dutch protocol in many real-world settings, while keeping HIPEC as a conditional option after CRS in selected patients.[6] In other words, the discussion evolved from “HIPEC yes/no” to “which HIPEC, for whom, in which center, with what expected risk.”
Patient selection: using PSDSS as a practical tool
Selection is often the highest-impact decision point. The Peritoneal Surface Disease Severity Score (PSDSS) has been evaluated in a large multi-institutional cohort and can stratify preoperative severity and prognosis using clinical and disease-extent variables.[7] In practice, tools like PSDSS help clinicians structure risk/benefit conversations, align expectations, and avoid impulsive indications—especially when imaging understages disease.
Center experience and expected morbidity: the “silent determinant”
CRS (with or without HIPEC) is complex. A decisive variable is program experience: selection, standardization, multidisciplinary care, and structured perioperative pathways. That is why suspected or confirmed peritoneal metastases should trigger early referral to an experienced PSM center, where indication, risk, and expectations can be aligned more precisely.[6]
If you want the “system-level” view of why structured pathways matter, here is a Peritoneo.life essay on value-based care in PSM:
https://peritoneo.life/value-based-care-peritoneal-surface-malignancies/
Practical checklists
For clinicians
Recognize that peritoneal disease is prognostically distinct and should not be managed as “generic metastasis.”[2] Assume CT may miss small-volume implants and underestimate true burden.[3] Avoid definitive decisions without structured staging and multidisciplinary discussion. When curative-intent is reasonable, prioritize the feasibility of complete cytoreduction.[4],[5] Interpret PRODIGE 7 correctly: CRS is the pillar; HIPEC depends on regimen and context.[5] Align the plan with contemporary consensus guidance on regimens.[6] Use tools like PSDSS to support selection and prognostic communication.[7] Refer early to an experienced PSM center.[6]
For patients and families
Peritoneal metastases require a strategic evaluation, ideally in an experienced PSM program.[6] Understand that imaging can underestimate small-volume disease.[3] Ask about treatment intent and whether a locoregional strategy is appropriate. Discuss risks and benefits with a team that performs CRS routinely. Seek a specialized second opinion if staging or eligibility is unclear.
Frequently asked questions
What are peritoneal metastases in colon cancer?
They are tumor implants on peritoneal surfaces within the abdomen. This pattern has distinct diagnostic and clinical behavior and may require a dedicated treatment strategy.[1]
Is “peritoneal carcinomatosis” the same as “peritoneal metastases”?
In everyday clinical language, the terms are often used interchangeably to describe tumor implants on the peritoneum.
Why is prognosis worse with peritoneal metastases?
Individual patient data analyses show that outcomes for isolated non-peritoneal metastases are better than for isolated peritoneal metastases, and peritoneal involvement drives major prognostic heterogeneity under systemic therapy.[2]
What symptoms can suggest peritoneal metastases?
Symptoms can be nonspecific: abdominal distension, discomfort/pain, changes in bowel habits, reduced appetite, weight loss and—when advanced—ascites or bowel obstruction. Early disease can be minimally symptomatic and may require careful staging.[1]
Does CT detect peritoneal metastases well?
Detection depends on size and location. CT can miss small implants; lesion-based sensitivity for very small disease can be low, and CT-PCI tends to underestimate true burden.[3]
How is proper staging performed?
Staging aims to estimate true peritoneal disease burden and eligibility for locoregional therapy. Because imaging can underestimate disease, experienced programs integrate imaging with clinical criteria and, when needed, direct assessment (such as diagnostic laparoscopy).[1],[3]
When is cytoreductive surgery indicated?
When complete macroscopic cytoreduction is realistically achievable with acceptable risk in a selected patient. Evidence across eras reinforces CRS as the foundation of curative-intent strategies in selected patients.[4],[5]
What is HIPEC, and why are protocols different?
HIPEC is heated intraperitoneal chemotherapy delivered during surgery. Benefit and risk depend on drug, dose, duration, and context. The modern question is not “HIPEC yes/no,” but “which HIPEC, for whom, and in which center.”[5],[6]
Did PRODIGE 7 eliminate HIPEC?
No. It showed no survival benefit for short-duration high-dose oxaliplatin HIPEC in that specific design while reinforcing the central value of high-quality CRS.[5] Consensus guidance then refined which regimens remain reasonable in selected settings.[6]
How do I stay updated on peritoneal oncology evidence?
You can subscribe to the Peritoneo.life newsletter for curated updates:
https://peritoneo.life/newsletter/
Conclusion
Peritoneal metastases in colon cancer represent a high-stakes scenario: worse outcomes under systemic therapy, higher risk of understaging, and decisions that demand precision.[2],[3] Peritoneal oncology has evolved—from landmark shifts toward CRS + HIPEC[4] to PRODIGE 7’s modern reframing that CRS quality and selection are the essential pillars, with HIPEC requiring regimen-specific judgment consistent with contemporary consensus.[5],[6]
Call to action
If you want to discuss this topic, share a case for educational purposes, or collaborate with Peritoneo.life, contact us here:
https://peritoneo.life/contact-us/
References
[1] Cortés-Guiral D, Hübner M, Alyami M, et al. Primary and metastatic peritoneal surface malignancies. Nat Rev Dis Primers. 2021;7(1):91. doi:10.1038/s41572-021-00326-6.
[2] Franko J, Shi Q, Goldman CD, et al. Prognosis of patients with peritoneal metastatic colorectal cancer given systemic therapy: an analysis of individual patient data from prospective randomised trials from the ARCAD database. Lancet Oncol. 2016;17(12):1709-1719. doi:10.1016/S1470-2045(16)30500-9.
[3] Sugarbaker PH. Preoperative assessment of cancer patients with peritoneal metastases for complete cytoreduction. Indian J Surg Oncol. 2016;7(2):130-136. doi:10.1007/s13193-016-0518-0.
[4] Verwaal VJ, van Ruth S, de Bree E, et al. Randomized trial of cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy and palliative surgery in patients with peritoneal carcinomatosis of colorectal cancer. J Clin Oncol. 2003;21(20):3737-3743. doi:10.1200/JCO.2003.04.187.
[5] Quénet F, Elias D, Roca L, et al. Cytoreductive surgery plus hyperthermic intraperitoneal chemotherapy versus cytoreductive surgery alone for colorectal peritoneal metastases (PRODIGE 7): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(2):256-266. doi:10.1016/S1470-2045(20)30599-4.
[6] Hübner M, et al. 2022 Peritoneal Surface Oncology Group International (PSOGI) Consensus on HIPEC regimens for peritoneal malignancies: colorectal cancer. Ann Surg Oncol. 2023. doi:10.1245/s10434-023-14368-5.
[7] Esquivel J, Lowy AM, Markman M, et al. The American Society of Peritoneal Surface Malignancies (ASPSM) Multiinstitution evaluation of the Peritoneal Surface Disease Severity Score (PSDSS) in patients with colorectal cancer with peritoneal carcinomatosis. Ann Surg Oncol. 2014;21(13):4195-4201. doi:10.1245/s10434-014-3798-z.